Chapter 2: The Chemical context of Life
Q8TYU
Page 28
Which coefficients must be placed in the following blanks so that all atoms are accounted for in the products?
\({C_6}{H_{12}}{O_6} \to \_\_\_{C_2}{H_6}{O_{}} + \_\_\_C{O_2}\)
(A)2,1
(B) 3,1
(C) 1,3
(D) 2,2
Q9TYU
Page 28
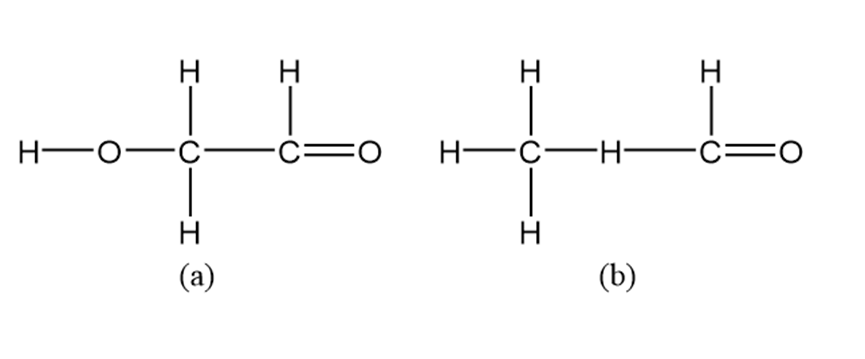
Draw Lewis dot structures for each hypothetical molecule shown below, using the correct number of valence electrons for each atom. Determine which molecules make sense because each atom has a complete valence shell, and each bond has the correct number of electrons. Explain what makes the other molecule nonsensical, considering the number of bonds each type of atom can make.