Chapter 18: Q38P (page 460)
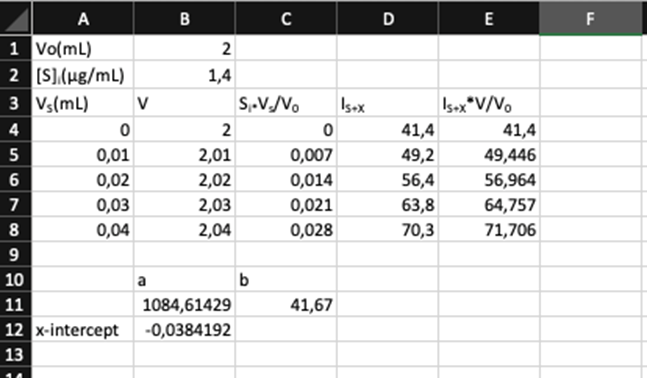
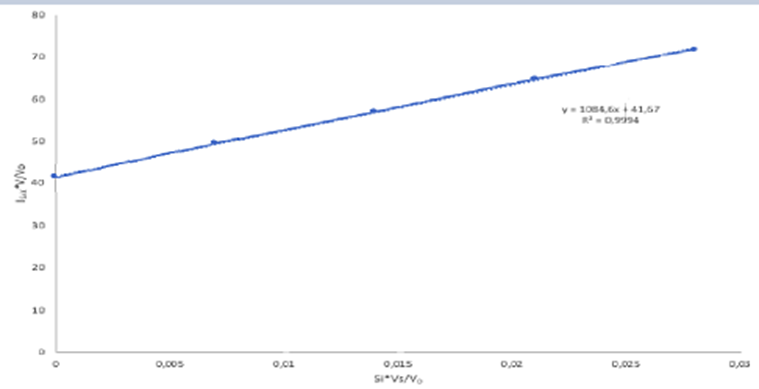
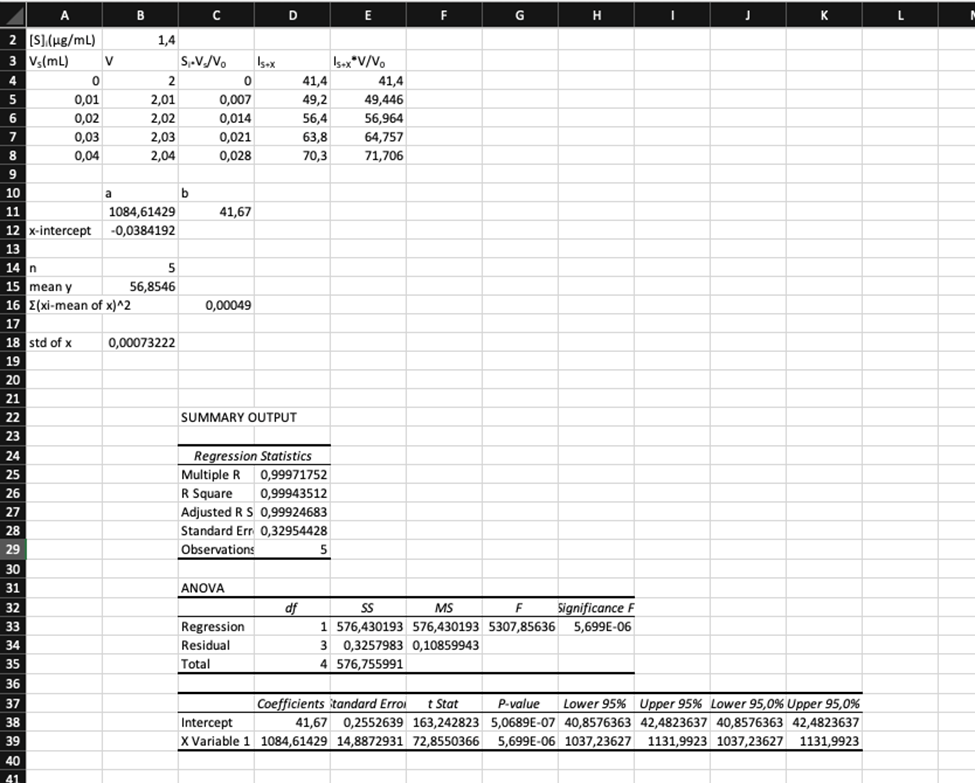
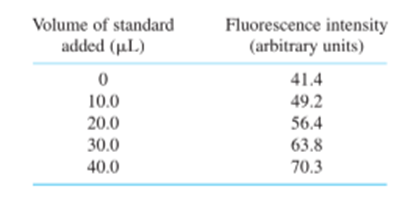
Standard addition. Selenium from 0.108 g of Brazil nuts was converted into the fluorescent product in Reaction 18-15, and extracted into 10.0 mL of cyclohexane. Then 2.00 mL of the cyclohexane solution were placed in a cuvet for fluorescence measurement. Standard additions of fluorescent product containingSe/mL are given in the table. Construct a standard addition graph to find the concentration of Se in the 2.00-mL unknown solution. Find the wt%of Se in the nuts and its uncertainty and 95% confidence interval
Short Answer
The interval is