Chapter 18: Q22P (page 458)
Nitrite ion, is a preservative for bacon and other foods, but it is potentially carcinogenic. A spectrophotometric determination ofmakes use of the following reactions
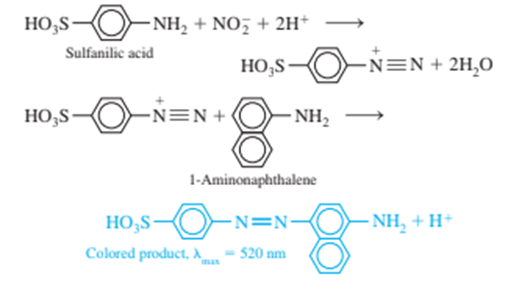
Here is an abbreviated procedure for the
determination:
1. To 50.0ml of unknown solution containing nitrite is added 1.00mL of sulfanilic acid solution.
2. After 10min , 2.00mL of 1 –a minonaphthalene solution and 1.00 mL of buffer are added.
3. After 15 min, the absorbance is read at 520 nm in a 5.00-cm cell.
The following solutions were analyzed:
A. 50.0 mL of food extract known to contain no nitrite (that is, a negligible amount); final absorbance =0.153.
B. 50.0mL of food extract suspected of containing nitrite; final absorbance \(=0.622\).
C. Same as B, but with added to the 50.0-mL sample; final absorbance =0.967.
(a) Calculate the molar absorptivity, of the colored product. Remember that a \(5.00\)-cm cell was used.
(b) How many micrograms ofwere present in 50.0mL of food extract?

