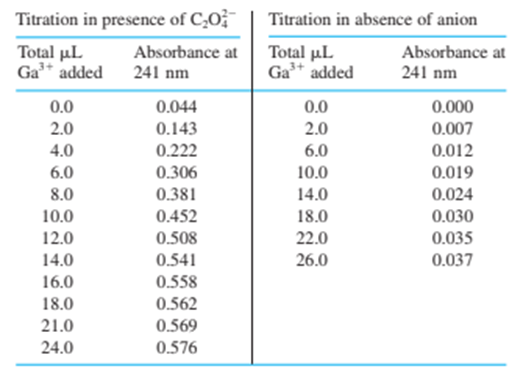
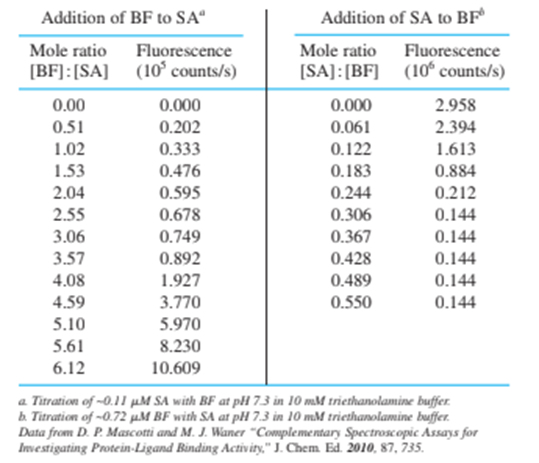
Chapter 18: QBE (page 455)
(a) A solution of compound A exhibited an absorbance of 0.624 at 238nmin a 1.000 cmcuvet; a blank solution containing only solvent had an absorbance of 0.029at the same wavelength. Find the molar absorptivity of compound A.
(b) The absorbance of an unknown solution of compound A in the same solvent and cuvet was 0.375 at 238 nm. Find the concentration of A in the unknown.
(c) A concentrated solution of compound A in the same solvent was diluted from an initial volume ofto a final volume of 25.00mLand then had an absorbance of 0.733. What is the concentration of A in the concentrated solution?
Short Answer
a) The molar absorptivity
b) The concentration of unknown A
c) Concentration of compound A is