Chapter 15: The Laws of Thermodynamics
Q60P
A “Carnot” refrigerator (the reverse of a Carnot engine) absorbs heat from the freezer compartment at a temperature of -17°C and exhausts it into the room at 25°C.
(a) How much work would the refrigerator do to change 0.65 kg of water at 25°C into ice at -17°C.
(b) If the compressor output is 105 W and runs 25% of the time, how long will this take?
Q61GP
It has been suggested that a heat engine could be developed that made use of the temperature difference between water at the surface of the ocean and water several hundred meters deep. In the tropics, the temperatures may be 27°C and 4°C, respectively.
(a) What is the maximum efficiency such an engine could have?
(b) Why might such an engine be feasible in spite of the low efficiency?
(c) Can you imagine any adverse environmental effects that might occur?
Q62GP
A cooling unit for a new freezer has an inner surface area of\({\bf{8}}{\bf{.0}}\;{{\bf{m}}{\bf{2}}}\), and is bounded by walls 12 cm thick with a thermal conductivity of\({\bf{0}}{\bf{.050}}\;{\bf{W/m}} \cdot {\bf{K}}\). The inside must be kept at -15°C in a room that is at 22°C. The motor for the cooling unit must run no more than 15% of the time. What is the minimum power requirement of the cooling motor?
Q63GP
Refrigeration units can be rated in “tons.” A 1-ton air conditioning system can remove sufficient energy to freeze 1 ton (2000 pounds = 909 kg) of 0°C water into 0°C ice in one 24-h day. If, on a 35°C day, the interior of a house is maintained at 22°C by the continuous operation of a 5-ton air conditioning system, how much does this cooling cost the homeowner per hour? Assume the work done by the refrigeration unit is powered by electricity that costs $0.10 per kWh and that the unit’s coefficient of performance is 18% that of an ideal refrigerator.\({\bf{1}}\;{\bf{kWh = 3}}{\bf{.60 \times 1}}{{\bf{0}}{\bf{6}}}\;{\bf{J}}\).
Q64GP
Two 1100 kg cars are traveling at a speed of\({\bf{85 km/h}}\)in opposite directions when they collide and are brought to rest. Estimate the change in entropy of the universe as a result of this collision. Assume\(T = {\bf{20\circ C}}\).
Q65GP
A 110 g insulated aluminum cup at 35°C is filled with 150 g of water at 45°C. After a few minutes, equilibrium is reached.
(a) Determine the final temperature and
(b) the total change in the entropy.
Q66GP
The burning of gasoline in a car releases about\({\bf{3}}{\bf{.0}} \times {\bf{1}}{{\bf{0}}{\bf{4}}}{\bf{ kcal/gal}}\).If a car averages\({\bf{41}}{\rm{ }}{\bf{km/gal}}\)when driving at a speed of\({\bf{110}}{\rm{ }}{\bf{km/h}}\),which requires 25 hp, what is the efficiency of the engine under those conditions?
Q67GP
A Carnot engine operates with\({T_L} = {\bf{20^\circ C}}\)and has an efficiency of 25%. By how many kelvins should the high operating temperature\({T_H}\)be increased to achieve an efficiency of 35%?
Q68GP
Calculate the work done by an ideal gas while going from state A to state C in Fig. 15–28 for each of the following processes:
(a) ADC,
(b) ABC, and
(c) AC directly.
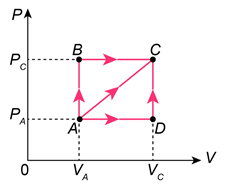
FIGURE 15–28
Problem 68
Q69GP
A 38% efficient power plant puts out 850 MW of electrical power. Cooling towers take away the exhaust heat.
(a) If the air \(\left( {{\bf{k}}{{\bf{m}}{\bf{3}}}} \right)\) temperature is allowed to rise by 7.0°C, estimate what volume of air is heated per day. Will the local climate be heated significantly?
(b) If the heated air were to form a layer 180 m thick, estimate how large an area it would cover for 24 h of operation. Assume the air has a density of \({\bf{1}}{\bf{.3 kg/}}{{\bf{m}}{\bf{3}}}\) and a specific heat of about \({\bf{1}}{\bf{.0}}{\rm{ }}{\bf{kJ/kg}} \cdot {\bf{\circ C}}\) at constant pressure.
