Chapter 2: Q10P (page 40)
Question: Draw the structures of the conjugate bases of the following acids:
a.
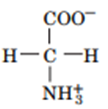
b.
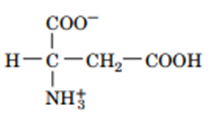
Short Answer
Answer
- The structure are:
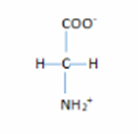
2. The structure are:
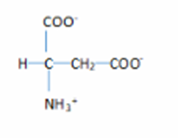
Learning Materials
Features
Discover
Chapter 2: Q10P (page 40)
Question: Draw the structures of the conjugate bases of the following acids:
a.

b.
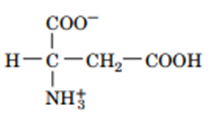
Answer

2. The structure are:

All the tools & learning materials you need for study success - in one app.
Get started for free
Use Coulomb’s law (Equation 2-1) to explain why a salt crystal such as NaCl remains intact in benzene (C6H6) but dissociates into ions in water.
Calculate the standard free energy change for the dissociation of HEPES.
You need a buffer at for use in purifying a protein at . You have chosen Tris, . You carefully make up Tris buffer, at data-custom-editor="chemistry" , and store it in the cold to equilibrate it to the temperature of the purification. When you measure the of the temperature-equilibrated buffer it has increased to . What is the explanation for this increase? How can you avoid this problem?
Calculate the pH of a 200 mL solution of pure water to which has been added 50 mL of 1 mM HCl.
Sketch a diagram of a water molecule and indicate the ends that bear partial positive and negative charges.
What do you think about this solution?
We value your feedback to improve our textbook solutions.